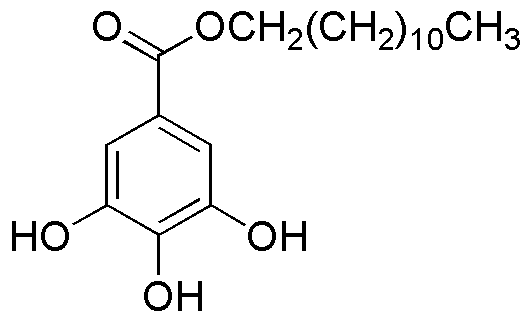
Lauryl gallate is widely utilized in research focused on:
- Food Preservation: This compound acts as an antioxidant, helping to extend the shelf life of various food products by preventing rancidity and oxidative damage.
- Cosmetic Formulations: Lauryl gallate is incorporated in skincare products for its ability to stabilize emulsions and enhance the product's texture, making it a popular choice in lotions and creams.
- Pharmaceutical Applications: It is used as an excipient in drug formulations, improving the stability and bioavailability of active ingredients, which is crucial for effective medication delivery.
- Polymer Industry: The compound serves as a processing aid in the production of plastics and coatings, enhancing their thermal stability and resistance to degradation.
- Research and Development: In laboratories, it is employed as a reagent in various chemical reactions, particularly in studies involving antioxidants and their effects on biological systems.
General Information
Properties
Safety and Regulations
Applications
Lauryl gallate is widely utilized in research focused on:
- Food Preservation: This compound acts as an antioxidant, helping to extend the shelf life of various food products by preventing rancidity and oxidative damage.
- Cosmetic Formulations: Lauryl gallate is incorporated in skincare products for its ability to stabilize emulsions and enhance the product's texture, making it a popular choice in lotions and creams.
- Pharmaceutical Applications: It is used as an excipient in drug formulations, improving the stability and bioavailability of active ingredients, which is crucial for effective medication delivery.
- Polymer Industry: The compound serves as a processing aid in the production of plastics and coatings, enhancing their thermal stability and resistance to degradation.
- Research and Development: In laboratories, it is employed as a reagent in various chemical reactions, particularly in studies involving antioxidants and their effects on biological systems.
Documents
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
Product Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Número de catálogo
Número de lote/lote
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.
Número de catálogo
Número de lote/lote
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
DownloadProduct Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
DownloadCertificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Número de catálogo
Número de lote/lote
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.