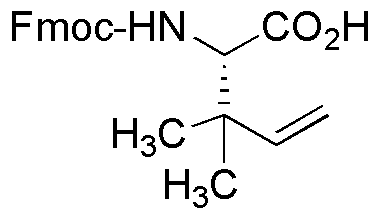
(S)-Fmoc-2-amino-3,3-dimethyl-pent-4-enoic acid is widely utilized in research focused on:
- Peptide Synthesis: This compound serves as a key building block in the synthesis of peptides, especially in solid-phase peptide synthesis, allowing for the creation of complex and functional peptide sequences.
- Drug Development: It plays a significant role in the pharmaceutical industry for developing novel therapeutics, particularly in designing peptide-based drugs that can target specific biological pathways.
- Bioconjugation: The compound is used in bioconjugation processes, where it helps attach biomolecules to surfaces or other molecules, enhancing the efficacy of diagnostics and drug delivery systems.
- Research in Protein Engineering: Researchers utilize this chemical to modify proteins, enabling the study of protein interactions and functions, which is crucial for understanding various biological processes.
- Functional Materials: It is also explored in the development of functional materials, such as hydrogels and nanomaterials, which have applications in tissue engineering and regenerative medicine.
General Information
Properties
Safety and Regulations
Applications
(S)-Fmoc-2-amino-3,3-dimethyl-pent-4-enoic acid is widely utilized in research focused on:
- Peptide Synthesis: This compound serves as a key building block in the synthesis of peptides, especially in solid-phase peptide synthesis, allowing for the creation of complex and functional peptide sequences.
- Drug Development: It plays a significant role in the pharmaceutical industry for developing novel therapeutics, particularly in designing peptide-based drugs that can target specific biological pathways.
- Bioconjugation: The compound is used in bioconjugation processes, where it helps attach biomolecules to surfaces or other molecules, enhancing the efficacy of diagnostics and drug delivery systems.
- Research in Protein Engineering: Researchers utilize this chemical to modify proteins, enabling the study of protein interactions and functions, which is crucial for understanding various biological processes.
- Functional Materials: It is also explored in the development of functional materials, such as hydrogels and nanomaterials, which have applications in tissue engineering and regenerative medicine.
Documents
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
Product Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Numéro de catalogue
Numéro de lot/série
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.
Numéro de catalogue
Numéro de lot/série
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
DownloadProduct Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
DownloadCertificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Numéro de catalogue
Numéro de lot/série
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.